In order to maintain the sterility of a sterile medical device, it is important that the device is stored in its sterile barrier system in a clean, dry environment and should not be subjected to excess temperature and should be inspected for any damage to its sterile barrier system before use.
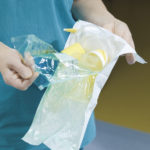
At the point of use, because only the inside surface of the pack is sterile, it is essential that the medical device does not touch the outer surface of the pack.
Sterile barrier systems are designed to ensure that this can easily be done by following the correct opening techniques. Packs are designed to allow the healthcare professional to hold a non- sterile part of the pack and peel the pack apart to present the medical device without touching it or allowing it to touch the outside of the pack. There are many techniques to achieve aseptic presentation of medical devices.
Devices should never be pushed through the pack. A pack which tears rather than peeling apart can compromise the sterility of the device by allowing loose fibres or other contaminates to touch the device.
Once the device is presented it should either be taken by a healthcare professional who has prepared themselves for a sterile procedure or dropped onto a sterile surface in a clean environment ready for immediate use.